  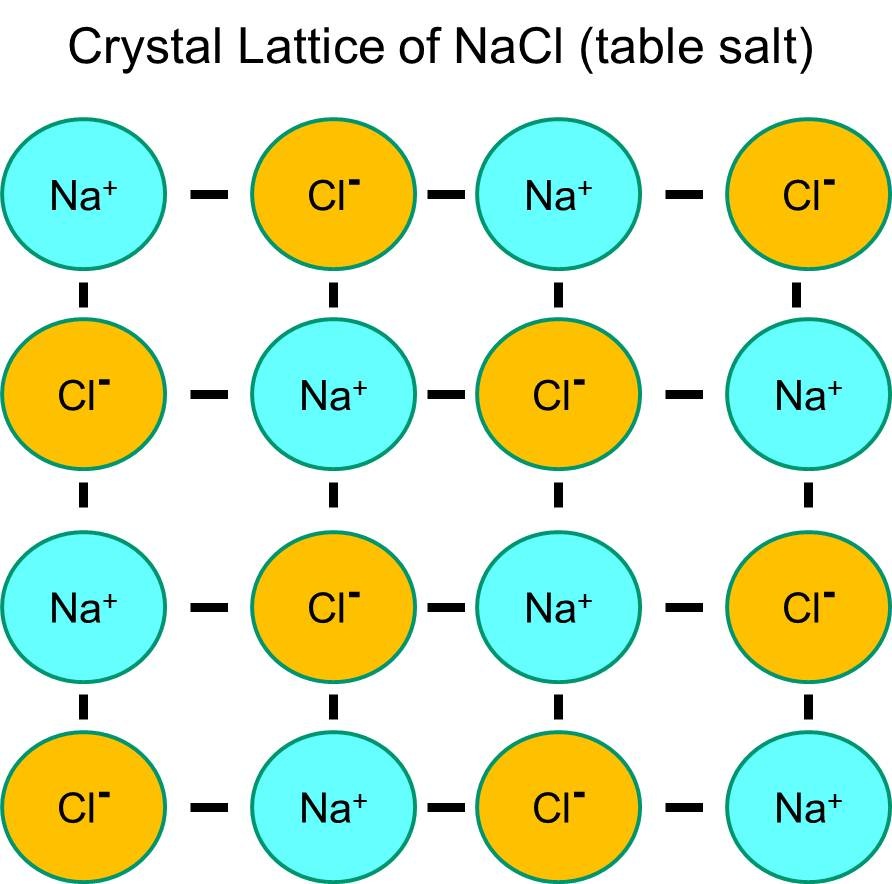
If Q 1 and Q 2 have opposite signs (as in NaCl, for example, where Q 1 is +1 for Na + and Q 2 is −1 for Cl −), then E is negative, which means that energy is released when oppositely charged ions are brought together from an infinite distance to form an isolated ion pair.Įnergy is always released when a bond is formed and correspondingly, it always requires energy to break a bond. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. In the example given, Q 1 = +1(1.6022 × 10 −19 C) and Q 2 = −1(1.6022 × 10 −19 C). An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. In this case, the proportionality constant, k, equals 8.999 × 109 J Best Answer Copy Ionic compounds are neutral because, even though they consist of positive and negative ions, the relative proportions between those ions are such that the total number of. Negative ions, or anions, are particles with one or more extra electrons, conferring a net negative charge to the particle. The ion has two electrons bound by the electromagnetic force to a nucleus containing one proton. In chemistry, this ion is called hydride. 
The hydrogen anion is an important constituent of the atmosphere of stars, such as the Sun. The equation can also be written using the charge of each ion, expressed in coulombs (C), incorporated in the constant. An air ioniser (or negative ion generator or Chizhevsky's chandelier) is a device that uses high voltage to ionise (electrically charge) air molecules. The hydrogen anion, H, is a negative ion of hydrogen, that is, a hydrogen atom that has captured an extra electron. This value of k includes the charge of a single electron (1.6022 × 10 −19 C) for each ion. The proportionality constant k is equal to 2.31 × 10 −28 J Where each ion’s charge is represented by the symbol Q. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
